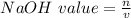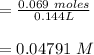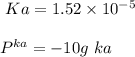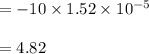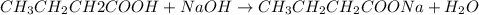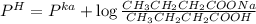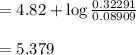Chemistry, 11.06.2020 17:57 lexiecooley
A 1.44 L buffer solution consists of 0.137 M butanoic acid and 0.275 M sodium butanoate. Calculate the pH of the solution following the addition of 0.069 moles of NaOH . Assume that any contribution of the NaOH to the volume of the solution is negligible. The Ka of butanoic acid is 1.52×10−5 .

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
What volume of a 2.00 m stock solution of naoh is needed to prepare 150. ml of 0.40 m solution?
Answers: 2

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
A 1.44 L buffer solution consists of 0.137 M butanoic acid and 0.275 M sodium butanoate. Calculate t...
Questions


History, 02.04.2020 22:32

Biology, 02.04.2020 22:32





Mathematics, 02.04.2020 22:32

Mathematics, 02.04.2020 22:32

Mathematics, 02.04.2020 22:32

English, 02.04.2020 22:32






Chemistry, 02.04.2020 22:32



Mathematics, 02.04.2020 22:32

 ".
".