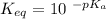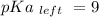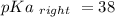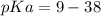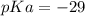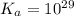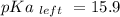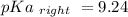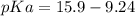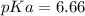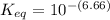Chemistry, 12.06.2020 04:57 katekayrodriguez10
Calculate Keq for these reactions and predict if the equilibrium will lie to the right or to the left as written. (You may enter your answer in scientific notation, e. g. 1.0*10^-9. Enter your answer to two significant figures.) Reaction 1: + + pKa = 9 pKa = 38 Keq = Equilibrium position = Reaction 2: + + pKa = 35 pKa = 25 Keq = Equilibrium position =

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 23.06.2019 01:00
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3

Chemistry, 23.06.2019 01:30
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
You know the right answer?
Calculate Keq for these reactions and predict if the equilibrium will lie to the right or to the lef...
Questions

Mathematics, 20.10.2020 21:01


Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01


Health, 20.10.2020 21:01

History, 20.10.2020 21:01

History, 20.10.2020 21:01

History, 20.10.2020 21:01

Law, 20.10.2020 21:01




Social Studies, 20.10.2020 21:01




Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01



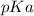 is mathematically evaluated as
is mathematically evaluated as 
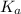 is mathematically evaluated as
is mathematically evaluated as