Chemistry, 11.06.2020 21:57 Kittylover65
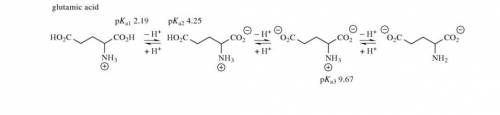
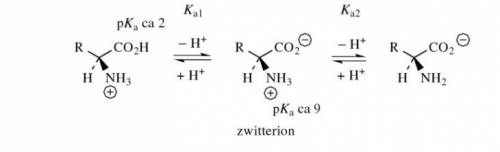
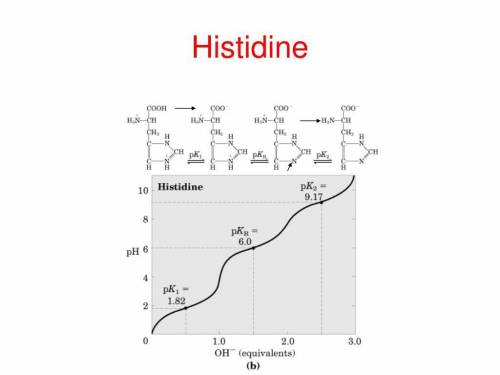
. The pI is called . The rule of calculating pI of an amino acid is that first, write the dissociation equation from fully protonated form to fully deprotonated form, label the charge of each form; second, identify the zwitterionic form (zero charge) and find the closest pKs (left and right side in the dissociation equation); third, average these two pKs. Write the dissociation equations for amino acids, glutamate, histidine, and calculate their pIs.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

You know the right answer?
. The pI is called . The rule of calculating pI of an amino acid is that first, write the dissociati...
Questions


Mathematics, 15.04.2020 04:33


Mathematics, 15.04.2020 04:33





Computers and Technology, 15.04.2020 04:33










English, 15.04.2020 04:33

Biology, 15.04.2020 04:34