

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Complete the following reactions using word and balanced equations including states. dilute phosphoric acid is added with a calcium hydroxide solution.
Answers: 1

Chemistry, 21.06.2019 20:10
Why is the vapor pressure of a warm lake higher than the vapor pressure of a cold lake? o a. warm water has a greater heat of vaporization. ob. warm water evaporates more quickly. cool water evaporates more quickly. od. cool water has a greater heat of vaporization.
Answers: 1

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2
You know the right answer?
According to the ideal gas law, what happens to the volume of a gas when the
temperature doubles (a...
Questions


Mathematics, 12.10.2020 23:01







Advanced Placement (AP), 12.10.2020 23:01



Mathematics, 12.10.2020 23:01



Mathematics, 12.10.2020 23:01


Mathematics, 12.10.2020 23:01

Chemistry, 12.10.2020 23:01

Mathematics, 12.10.2020 23:01

Mathematics, 12.10.2020 23:01

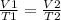 And volume and temperature are directly correlated.
And volume and temperature are directly correlated.

