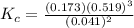Chemistry, 10.06.2020 18:57 ninaaforever
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studying this reaction fills a tank with of ammonia gas, and when the mixture has come to equilibrium measures the amount of nitrogen gas to be 13. mol. Calculate the concentration equilibrium constant for the decomposition of ammonia at the final temperature of the mixture.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 07:00
Scuba divers use tanks of compressed air to them breathe. gases can be compressed because?
Answers: 1
You know the right answer?
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studyin...
Questions

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00





Chemistry, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00


English, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00


Business, 24.02.2021 01:00

Biology, 24.02.2021 01:00

Computers and Technology, 24.02.2021 01:00



English, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

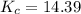
 ↔
↔ 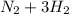
![[NH_3] = \frac{n_1}{V_1} = \frac{29}{75}](/tpl/images/0681/9995/b151c.png)
![[NH_3] = 0.387 \ M](/tpl/images/0681/9995/5ee5d.png)
![[N_2] = \frac{n_2}{V_2}](/tpl/images/0681/9995/a9b63.png)
![[N_2] = 0.173 \ M](/tpl/images/0681/9995/f89ee.png)
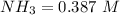

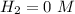
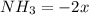 (this implies that it losses two moles of concentration )
(this implies that it losses two moles of concentration )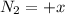 (this implies that it gains 1 moles)
(this implies that it gains 1 moles) (this implies that it gains 3 moles)
(this implies that it gains 3 moles)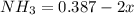
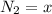
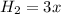
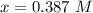
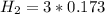

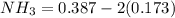
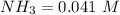
![K_c = \frac{[N_2][H_2]^3}{[NH_3]^2}](/tpl/images/0681/9995/10463.png)