
Chemistry, 09.06.2020 03:57 tonimgreen17p6vqjq
The chemical formula of a compound describes the elements the compound contains and the
A. pH of the compound.
B. conduction of the compound.
C. ratio of elements in the compound.
D. ratio of neutrons to protons in each element.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 23.06.2019 04:30
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1
You know the right answer?
The chemical formula of a compound describes the elements the compound contains and the
A. pH of th...
Questions


Mathematics, 20.05.2021 01:30

Mathematics, 20.05.2021 01:30


Chemistry, 20.05.2021 01:30



Social Studies, 20.05.2021 01:30

Mathematics, 20.05.2021 01:30

English, 20.05.2021 01:30



Mathematics, 20.05.2021 01:30

Mathematics, 20.05.2021 01:30



Mathematics, 20.05.2021 01:30


Social Studies, 20.05.2021 01:30

English, 20.05.2021 01:30

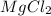 is the chemical formula for Magnesium chloride and it shows that the ratio of Magnesium to Chlorine (Cl) is 1:2.
is the chemical formula for Magnesium chloride and it shows that the ratio of Magnesium to Chlorine (Cl) is 1:2.

