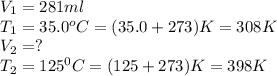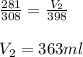Chemistry, 08.06.2020 09:57 brucewayne8499
Carbon dioxide gas at a temperature of 35.0 C and a volume of 281mL. Determine the volume of the gas when the temperature of the gas is raised to 125 C if the pressure remains constant.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
This is a characteristic of the elements in the periodic table that shows a pattern. it may increase or decrease across or down the table.
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1

Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3
You know the right answer?
Carbon dioxide gas at a temperature of 35.0 C and a volume of 281mL. Determine the volume of the gas...
Questions


Mathematics, 22.10.2020 09:01

Mathematics, 22.10.2020 09:01



Chemistry, 22.10.2020 09:01


Social Studies, 22.10.2020 09:01

Mathematics, 22.10.2020 09:01

English, 22.10.2020 09:01

Mathematics, 22.10.2020 09:01


Biology, 22.10.2020 09:01



Mathematics, 22.10.2020 09:01

English, 22.10.2020 09:01



Mathematics, 22.10.2020 09:01

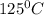 is 363 ml
is 363 ml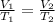
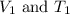 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.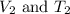 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.