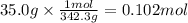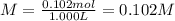Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1

Chemistry, 23.06.2019 14:00
[07.06] which of the following chemical reactions is an oxidation-reduction reaction? (2 points) wo3 + 3h2 yields w + 3h2o kno3 + licl yields lino3 + kcl caso4 + 2nacl yields na2so4 + cacl2 mg(no3)2 + 2hbr yields mgbr2 + 2hno3
Answers: 1

Chemistry, 23.06.2019 15:30
Consider these four line graphs representing speed in meters/second, where each x-axis is labelled in seconds and each y-axis is labelled in meters. which line graph indicates the greatest speed at 5 seconds?
Answers: 1
You know the right answer?
u make 1.000 L of an aqueous solution that contains 35.0 g of sucrose (C12H22O11).Part AWhat is the...
Questions



Mathematics, 07.05.2020 14:00






Biology, 07.05.2020 14:00

Mathematics, 07.05.2020 14:00







Mathematics, 07.05.2020 14:00

Physics, 07.05.2020 14:00