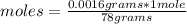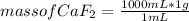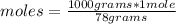In the background information, it was stated that CaF2 has solubility, at room temperature, of 0.00160 g per 100 g of water. How many moles of CaF2 can dissolve in 100 g of water? If the density of a saturated solution of CaF2 is 1.00 g/mL, how many moles of CaF2 will dissolve in exactly 1.00 L of solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3

Chemistry, 23.06.2019 01:30
Witch two conditions can limit the usefulness of the kinetic molecular theory in describing gas behavior?
Answers: 2

Chemistry, 23.06.2019 09:20
Description: biological systems 5 differ from chemical systems when it comes to equilibrium. in a chemical system once equilibrium is met, no other reactions occur. in a biological system, a dynamic equilibrium is used when a substrate is turned into a product, another reaction creates the same substrate thus keeping the concentrations stagnant. this allows for cells to continually make new compounds without messing the delta g for the systems instructions: write a response to the following prompt and then respond to your peers. your individual response is due thursday at midnight (cst) your response to your peers is due saturday at midnight (cst) prompt: propose what would happen if a living cell all the sudden reached chemical equilibrium. also discuss the effects of build up of a particular substrate on a biological system. how would this affect overall delta g's?
Answers: 3
You know the right answer?
In the background information, it was stated that CaF2 has solubility, at room temperature, of 0.001...
Questions




Arts, 21.05.2020 21:15



Mathematics, 21.05.2020 21:15

Mathematics, 21.05.2020 21:15



Biology, 21.05.2020 21:15


Mathematics, 21.05.2020 21:15


Mathematics, 21.05.2020 21:15



English, 21.05.2020 21:15


Mathematics, 21.05.2020 21:15