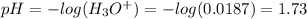A 1-liter solution contains 0.494 M hydrofluoric acid and 0.371 M potassium fluoride. Addition of 0.408 moles of hydrochloric acid will: (Assume that the volume does not change upon the addition of hydrochloric acid.)
a. Raise the pH slightly
b. Lower the pH slightly
c. Raise the pH by several units
d. Lower the pH by several units
e. Not change the pH
f. Exceed the buffer capacity

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The p sub shell can hold up to 8 electrons in an atom. true or false?
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2


Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
A 1-liter solution contains 0.494 M hydrofluoric acid and 0.371 M potassium fluoride. Addition of 0....
Questions

Mathematics, 01.12.2019 03:31

Social Studies, 01.12.2019 03:31


Health, 01.12.2019 03:31


English, 01.12.2019 03:31

History, 01.12.2019 03:31

Arts, 01.12.2019 03:31

History, 01.12.2019 03:31


Mathematics, 01.12.2019 03:31


Mathematics, 01.12.2019 03:31


History, 01.12.2019 03:31

Mathematics, 01.12.2019 03:31


Biology, 01.12.2019 03:31

Mathematics, 01.12.2019 03:31

![pH = pKa + log(\frac{[KF]}{[HF]})](/tpl/images/0677/3728/a79c6.png)
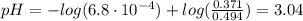
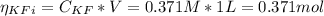
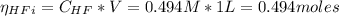
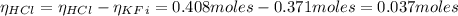
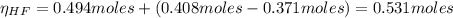
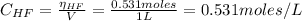
![Ka = \frac{[H_{3}O^{+}][F^{-}]}{[HF]}](/tpl/images/0677/3728/2de73.png)
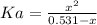 (2)
(2)