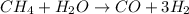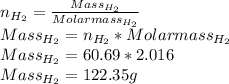Chemistry, 04.06.2020 13:30 zaylencollins55
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen. Suppose a chemical engineer studying a new catalyst for the reform reaction finds that liters per second of methane are consumed when the reaction is run at and . Calculate the r

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 01:30
In which phase of mitosis do the spindle fibers pull the chromosomes apart to opposite sides of the cell ?
Answers: 1
You know the right answer?
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carb...
Questions

Mathematics, 22.05.2021 07:10


English, 22.05.2021 07:10


Advanced Placement (AP), 22.05.2021 07:10


English, 22.05.2021 07:10

Mathematics, 22.05.2021 07:10

Biology, 22.05.2021 07:10


Biology, 22.05.2021 07:10

Mathematics, 22.05.2021 07:10

Mathematics, 22.05.2021 07:20

Mathematics, 22.05.2021 07:20

Mathematics, 22.05.2021 07:20

English, 22.05.2021 07:20

Mathematics, 22.05.2021 07:20

English, 22.05.2021 07:20

Arts, 22.05.2021 07:20