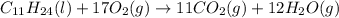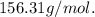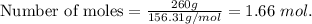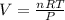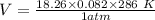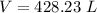Combustion of hydrocarbons such as undecane (C_11H_24) produces carbon dioxide, a "greenhouse gas." Greenhouse gases in the Earth's atmosphere can trap the Sun's heat, raising the average temperature of the Earth. For this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.
1. Write a balanced chemical equation, including physical state symbols, for the combustion of liquid undecane into gaseous carbon dioxide and gaseous water.
2. Suppose 0.260 kg of undecane are burned in air at a pressure of exactly 1 atm and a temperature of 13.0°C. Calculate the volume of carbon dioxide gas that is produced. Be sure your answer has the correct number of significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1

Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1


Chemistry, 23.06.2019 06:40
15. what volume of cci, (d = 1.6 g/cc) contain6.02 x 1025 cci, molecules (ci = 35.5)(1) 10.5 l(2) 250 ml(3) 9.625 l(4) 1.712 lplz answer with step by step explanation
Answers: 1
You know the right answer?
Combustion of hydrocarbons such as undecane (C_11H_24) produces carbon dioxide, a "greenhouse gas."...
Questions

Mathematics, 08.07.2019 07:00






Mathematics, 08.07.2019 07:00


History, 08.07.2019 07:00



Chemistry, 08.07.2019 07:00


Mathematics, 08.07.2019 07:00

Mathematics, 08.07.2019 07:00

Chemistry, 08.07.2019 07:00

Mathematics, 08.07.2019 07:00

Mathematics, 08.07.2019 07:00

Biology, 08.07.2019 07:00

Mathematics, 08.07.2019 07:00