
Chemistry, 31.05.2020 03:00 cookiemonster0476
Hard water often contains dissolved Ca2+ and Mg2+ ions. One way to soften water is to add phosphates. The phosphate ion forms insoluble precipitates with calcium and magnesium ions, removing them from solution. Suppose that a solution is 0.054 M in calcium chloride and 0.093 M in magnesium nitrate. What mass of sodium phosphate would have to be added to 1.4 L of this solution to completely eliminate the hard water ions? Assume complete reaction. Enter a numerical answer only, in terms of grams to two significant figures.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1


Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
Hard water often contains dissolved Ca2+ and Mg2+ ions. One way to soften water is to add phosphates...
Questions



Health, 26.10.2019 11:43

Biology, 26.10.2019 11:43




Mathematics, 26.10.2019 11:43


Mathematics, 26.10.2019 11:43


Arts, 26.10.2019 11:43




Mathematics, 26.10.2019 11:43



Mathematics, 26.10.2019 11:43

Biology, 26.10.2019 11:43

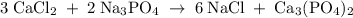
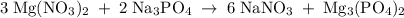
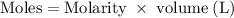
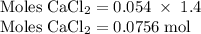
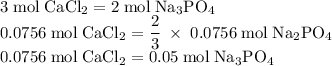
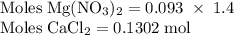
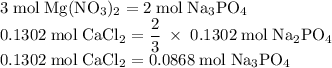
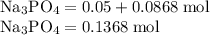
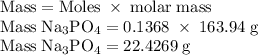
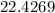 grams of sodium phosphate must be added to 1.4 L of this solution to completely eliminate the hard water ions
grams of sodium phosphate must be added to 1.4 L of this solution to completely eliminate the hard water ions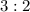
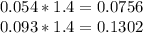
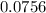 mole of CaCl2 is equal to
mole of CaCl2 is equal to 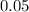 Na3PO4
Na3PO4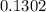 mole of CaCl2 is equal to
mole of CaCl2 is equal to 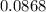 Na3PO4
Na3PO4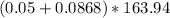 g/mol
g/mol 

