What is the mass in grams of 1.50 X 1023 molecules of NaOH?
a. 10.0 g
b. 20.0 g
...

Chemistry, 29.05.2020 05:57 fruitbutt77
What is the mass in grams of 1.50 X 1023 molecules of NaOH?
a. 10.0 g
b. 20.0 g
c. 30.0 g
d. 40.0 g
e. 60.0 g

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Type the correct answer in the box. spell all words correctly.what is the correct term for living the most sustainable life you can within your current circumstances? when your are being as sustainable as you can within your current lifestyle, you are said to be sustainability.
Answers: 3

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1
You know the right answer?
Questions


Spanish, 26.02.2020 23:27




History, 26.02.2020 23:27








History, 26.02.2020 23:27






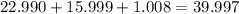 . But before we do anything with this number we must first convert
. But before we do anything with this number we must first convert  molecules to moles. We can do this by dividing by Avogadro's number or
molecules to moles. We can do this by dividing by Avogadro's number or 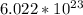 (
(  moles). We then convert moles to grams by multiplying by the molar mass of NaOH, (
moles). We then convert moles to grams by multiplying by the molar mass of NaOH, (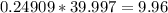 grams). This is almost exactly 10 and the margin of error is likely due to the original numbers not being precise enough.
grams). This is almost exactly 10 and the margin of error is likely due to the original numbers not being precise enough. 

