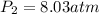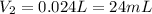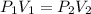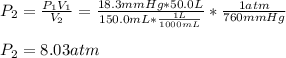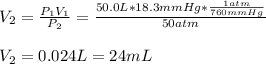Chemistry, 29.05.2020 03:02 Franklyn3834
3. A 50.0 L sample of gas collected in the upper atmosphere at a pressure of 18.3 mmHg is compressed into a 150.0 mL container at the same temperature.
a. What is the new pressure, in atm?
b. To what volume would the original sample have had to be compressed to exert a pressure of 10.0 atm?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
You know the right answer?
3. A 50.0 L sample of gas collected in the upper atmosphere at a pressure of 18.3 mmHg is compressed...
Questions

Mathematics, 15.07.2021 21:20

English, 15.07.2021 21:20



Computers and Technology, 15.07.2021 21:20


Physics, 15.07.2021 21:20

Mathematics, 15.07.2021 21:20

Mathematics, 15.07.2021 21:20

Mathematics, 15.07.2021 21:20


Biology, 15.07.2021 21:20






Mathematics, 15.07.2021 21:20

Mathematics, 15.07.2021 21:20