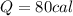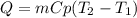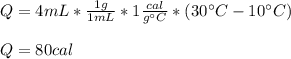Calculate the amount of heat needed to raise the temperature of 4 ml of water from 10C to 30C
...

Chemistry, 28.05.2020 20:57 juansantos7b
Calculate the amount of heat needed to raise the temperature of 4 ml of water from 10C to 30C
1 point
40 calories
80 calories
120 calories
1200 calories

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2
You know the right answer?
Questions




SAT, 20.10.2021 14:00






SAT, 20.10.2021 14:00

Mathematics, 20.10.2021 14:00

English, 20.10.2021 14:00


Mathematics, 20.10.2021 14:00

English, 20.10.2021 14:00


Health, 20.10.2021 14:00


Mathematics, 20.10.2021 14:00