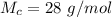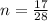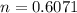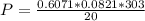Chemistry, 27.05.2020 16:57 urstruulyemily
A reaction between liquid reactants takes place at in a sealed, evacuated vessel with a measured volume of . Measurements show that the reaction produced of carbon monoxide gas. Calculate the pressure of carbon monoxide gas in the reaction vessel after the reaction. You may ignore the volume of the liquid reactants. Be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3
You know the right answer?
A reaction between liquid reactants takes place at in a sealed, evacuated vessel with a measured vol...
Questions





English, 04.08.2019 04:00


History, 04.08.2019 04:00


Mathematics, 04.08.2019 04:00






Mathematics, 04.08.2019 04:00

English, 04.08.2019 04:00




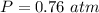
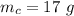
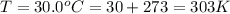
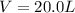
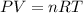
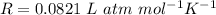
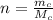
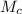 is the molar mass of carbon monoxide which is a constant with value
is the molar mass of carbon monoxide which is a constant with value