
Chemistry, 27.05.2020 04:58 phsycotic121
What is the concentration of the silver ion in silver chromate, Ag₂CrO₄, if its solubility product constant (Kₛₚ) is 1.2 x 10⁻¹². Hint: write the equation first! *
2 points
1.4 x 10⁻⁵
1.1 x 10⁻⁹
1.6 x 10⁻¹²
2.4 x 10⁻¹²

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:40
Asingle atom of an element has 21 neutrons, 20 electrons, and 20 protons. which element is it? ok o z
Answers: 1

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

You know the right answer?
What is the concentration of the silver ion in silver chromate, Ag₂CrO₄, if its solubility product c...
Questions


English, 20.03.2020 10:15




Geography, 20.03.2020 10:15








Computers and Technology, 20.03.2020 10:15





History, 20.03.2020 10:15

Mathematics, 20.03.2020 10:15

![[Ag^+]=1.3x10^{-4}M](/tpl/images/0666/7571/0a833.png)
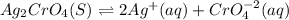
![Ksp=[Ag]^2[CrO_4^{-2}]](/tpl/images/0666/7571/75fed.png)
 due to the dissolution of silver chromate, we obtain:
due to the dissolution of silver chromate, we obtain: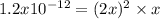
![x=\sqrt[3]{\frac{1.2x10^{-12}}{2^2} } = 6.7x10^{-5}M](/tpl/images/0666/7571/1e8f3.png)
![[Ag^+]=2x=2*6.7x10^{-5}M=1.3x10^{-4}M](/tpl/images/0666/7571/1be44.png)


