
Chemistry, 26.05.2020 05:57 enrique3300
What is the pH of a solution of KOH with a hydroxide concentration of [OH⁻] = 1.10 x 10⁻⁴

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3
You know the right answer?
What is the pH of a solution of KOH with a hydroxide concentration of [OH⁻] = 1.10 x 10⁻⁴...
Questions

Chemistry, 06.05.2020 23:57

Mathematics, 06.05.2020 23:57


Mathematics, 06.05.2020 23:57


Mathematics, 06.05.2020 23:57

Mathematics, 06.05.2020 23:57

Mathematics, 06.05.2020 23:57


History, 06.05.2020 23:57



Biology, 06.05.2020 23:57


Computers and Technology, 06.05.2020 23:57




Spanish, 06.05.2020 23:58

Mathematics, 06.05.2020 23:58

![p[H+] = 10.042](/tpl/images/0665/7150/22132.png)
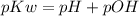 ......eq (1)
......eq (1)![= - log [1.10 * 10^{-4}]](/tpl/images/0665/7150/d0c85.png)
![= - [-3.958]\\= 3.958](/tpl/images/0665/7150/381e4.png)
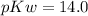
![14.0 = p[H+] + 3.958\\p[H+] = 14 - 3.958\\p[H+] = 10.042](/tpl/images/0665/7150/4fd37.png)


