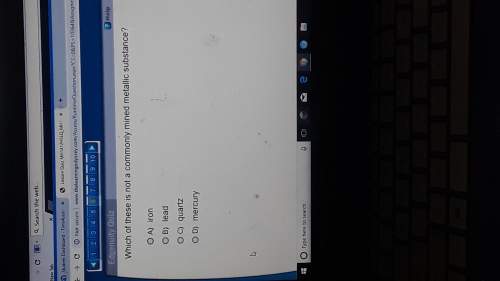
What type of chemical reaction is shown below?*
H2CO3 (aq)
+ H2O
(1)
+ CO2 (...

Chemistry, 24.05.2020 06:57 wasscrackin
What type of chemical reaction is shown below?*
H2CO3 (aq)
+ H2O
(1)
+ CO2 (g)
combustion
O synthesis
decomposition
single replacement
double replacement

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

You know the right answer?
Questions

English, 29.08.2020 07:01

Mathematics, 29.08.2020 07:01



Mathematics, 29.08.2020 08:01


Mathematics, 29.08.2020 08:01


English, 29.08.2020 08:01




Mathematics, 29.08.2020 08:01




Mathematics, 29.08.2020 08:01


Computers and Technology, 29.08.2020 08:01