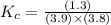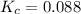Chemistry, 23.05.2020 21:58 melinda12ms
In the reaction NO + NO2 ⇌ N2O3, an experiment finds equilibrium concentrations of [NO] = 3.8 M, [NO2] = 3.9 M, and [N2O3] = 1.3 M. What is the equilibrium constant Kc for this reaction?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2
You know the right answer?
In the reaction NO + NO2 ⇌ N2O3, an experiment finds equilibrium concentrations of [NO] = 3.8 M, [NO...
Questions

Mathematics, 09.04.2021 02:40


Biology, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40

History, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40



Mathematics, 09.04.2021 02:40


English, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40

Mathematics, 09.04.2021 02:40



Mathematics, 09.04.2021 02:40


Health, 09.04.2021 02:40

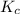 for this reaction is 0.088
for this reaction is 0.088
![K_c=\frac{[N_2O_3]}{[NO_2][NO]}](/tpl/images/0663/9574/d5725.png)