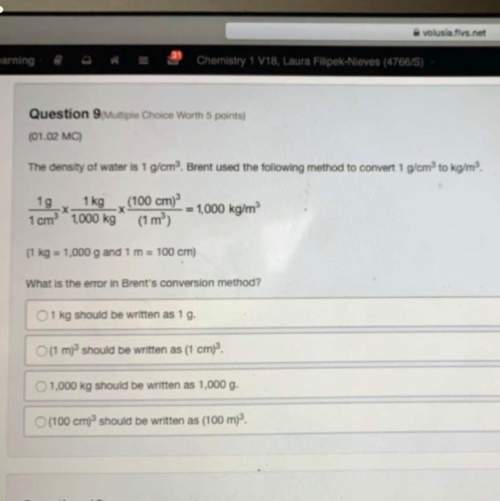
The balanced equation for combustion in an acetylene torch is shown below:
2C2H2 + 5O2 → 4CO2...

Chemistry, 23.05.2020 05:00 rileyallen4186pd5tgy
The balanced equation for combustion in an acetylene torch is shown below:
2C2H2 + 5O2 → 4CO2 + 2H2O
The acetylene tank contains 35.0 mol C2H2, and the oxygen tank contains 84.0 mol O2.
How many moles of CO2 are produced when 35.0 mol C2H2 react completely? ⇒ 70.0 mol CO2
How many moles of CO2 are produced when 84.0 mol O2 completely react? mol CO2

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 23.06.2019 08:30
Sand is more likely than shale to preserve fossils. true false
Answers: 2

Chemistry, 23.06.2019 12:10
What is the correct name for hg(no3)2? mercury (i) nitrate mercury (ii) nitrate mercury nitroxide mercury dinitride
Answers: 1

Chemistry, 23.06.2019 23:20
An unknown organic compound (0.315 g) containing only c, h, and o produces 0.771 of co2 and 0.105 g of h2o when it undergoes complete combustion. the approximate molar mass is 108 g/mol. which of the following compounds could be the identification of the unknown?
Answers: 2
You know the right answer?
Questions

Mathematics, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14

English, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14






Mathematics, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14

History, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14


Mathematics, 29.01.2020 00:14

Mathematics, 29.01.2020 00:14



Mathematics, 29.01.2020 00:40