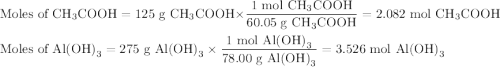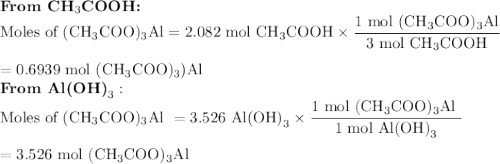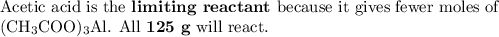Chemistry, 23.05.2020 02:06 elizabethhubbe
Quien y en que cantidad sera el reactivo limite, si utilizamos 125 g de ácido, H (CH3COO) y 275 g de hidróxido , Al(OH)3

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Quien y en que cantidad sera el reactivo limite, si utilizamos 125 g de ácido, H (CH3COO) y 275 g de...
Questions



Biology, 08.01.2020 21:31


Mathematics, 08.01.2020 21:31



English, 08.01.2020 21:31



Chemistry, 08.01.2020 21:31



Mathematics, 08.01.2020 21:31

Chemistry, 08.01.2020 21:31

Computers and Technology, 08.01.2020 21:31


English, 08.01.2020 21:31