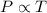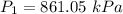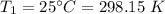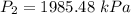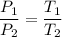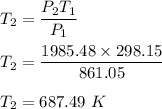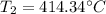Chemistry, 22.05.2020 23:57 naimareiad
2. A sample of gas has a pressure of 861.05 kPa at a temperature of 25°C. What temperature is required to
increase the pressure to 1985.48 kPa?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 07:30
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1
You know the right answer?
2. A sample of gas has a pressure of 861.05 kPa at a temperature of 25°C. What temperature is requir...
Questions


History, 30.09.2019 03:50

Mathematics, 30.09.2019 03:50

Social Studies, 30.09.2019 03:50



History, 30.09.2019 03:50



Chemistry, 30.09.2019 03:50

Mathematics, 30.09.2019 03:50

Physics, 30.09.2019 03:50

History, 30.09.2019 03:50




History, 30.09.2019 03:50

Physics, 30.09.2019 03:50