PLEAS HEL I BE FOR HEL
How many molecules are in a sample of water, H2O, with a mass of...

Chemistry, 22.05.2020 18:58 kokilavani
PLEAS HEL I BE FOR HEL
How many molecules are in a sample of water, H2O, with a mass of 63.00 grams?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
Questions



Mathematics, 11.02.2021 16:50




Mathematics, 11.02.2021 16:50

Chemistry, 11.02.2021 16:50

Chemistry, 11.02.2021 16:50

Biology, 11.02.2021 16:50

Mathematics, 11.02.2021 16:50


Mathematics, 11.02.2021 16:50


Mathematics, 11.02.2021 16:50

Mathematics, 11.02.2021 16:50


Mathematics, 11.02.2021 16:50


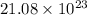 molecules in 63.00 g of
molecules in 63.00 g of 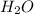
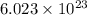 of particles.
of particles.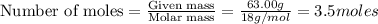
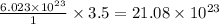 molecules.
molecules.

