
Chemistry, 22.05.2020 01:00 ayoismeisjjjjuan
At high temperatures, calcium carbonate (CaCO3) decomposes to form calcium oxide (CaO) and carbon dioxide (CO2) gas. The average mass of 1 calcium oxide molecule is 56.079 amu, and the average mass of 1 carbon dioxide molecule is 44.009 amu.
CaCO3 CaO + CO2
Which is the total mass of the 1 calcium carbonate molecule?
A. 12.070 amu
B. 44.009 amu
C. 56.079 amu
D. 100.088 amu

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
At high temperatures, calcium carbonate (CaCO3) decomposes to form calcium oxide (CaO) and carbon di...
Questions

Mathematics, 09.01.2020 00:31






Chemistry, 09.01.2020 00:31






Biology, 09.01.2020 00:31

History, 09.01.2020 00:31

Social Studies, 09.01.2020 00:31



Physics, 09.01.2020 00:31

Geography, 09.01.2020 00:31

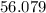 amu
amu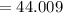 amu
amu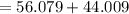
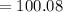 amu
amu

