
Chemistry, 21.05.2020 22:08 rsimmons696
The ph of an aqueous solution at 25.0°c is 10.66. what is the molarity of h+ in this solution? the ph of an aqueous solution at 25.0°c is 10.66. what is the molarity of h+ in this solution? 4.6 à 10-4 3.3 2.2 à 10-11 1.1 à 10-13 4.6 à 1010

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1
You know the right answer?
The ph of an aqueous solution at 25.0°c is 10.66. what is the molarity of h+ in this solution? the...
Questions



Biology, 05.07.2019 12:40

English, 05.07.2019 12:40




Chemistry, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40



English, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40



Computers and Technology, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40

History, 05.07.2019 12:40


Mathematics, 05.07.2019 12:40

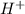 in this solution is
in this solution is 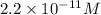
![pH=-\log [H^+]](/tpl/images/0659/9285/37e81.png)
![10.66=-\log[H^+]](/tpl/images/0659/9285/4a0df.png)
![[H^+]=10^{-10.66}](/tpl/images/0659/9285/0b8ff.png)
![[H^+]=2.2\times 10^{-11}](/tpl/images/0659/9285/b8988.png)


