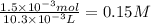Chemistry, 21.05.2020 05:00 cpratt2558
The student titrated 10 ml of standered 0.15 M HCl with his sodium hydroxide solution. When the titration reached the equivalence point, the student found that he had used
10.3 ml of Sodium hydroxide solution. Calculate the molarity of the sodium hydroxide solution.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is lincoln's purpose in writing this speech? question 1 options: to stress the difficulties of war to honor those who died in the war to call for an end to the war to call the country to join a new war
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
The student titrated 10 ml of standered 0.15 M HCl with his sodium hydroxide solution. When the titr...
Questions

Mathematics, 18.07.2019 18:40

History, 18.07.2019 18:40

Social Studies, 18.07.2019 18:40

Mathematics, 18.07.2019 18:40


History, 18.07.2019 18:40




Business, 18.07.2019 18:40

Mathematics, 18.07.2019 18:40


Biology, 18.07.2019 18:40



History, 18.07.2019 18:40

History, 18.07.2019 18:40

Mathematics, 18.07.2019 18:40

Social Studies, 18.07.2019 18:40