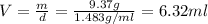Chemistry, 19.05.2020 14:58 Demarcusclincy
Chloroform is a commonly used anesthetic with a density of 1.483 g/mL. Determine the volume of chloroform needed to deliver a 9.37 g sample of the anesthetic

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Chloroform is a commonly used anesthetic with a density of 1.483 g/mL. Determine the volume of chlor...
Questions

Mathematics, 27.11.2020 19:10

Biology, 27.11.2020 19:10


Mathematics, 27.11.2020 19:10


Mathematics, 27.11.2020 19:10


Computers and Technology, 27.11.2020 19:20

Business, 27.11.2020 19:20



English, 27.11.2020 19:20

Chemistry, 27.11.2020 19:20

English, 27.11.2020 19:20

Geography, 27.11.2020 19:20

Biology, 27.11.2020 19:20

Social Studies, 27.11.2020 19:20



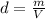 ⇒
⇒