Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:40
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 21.06.2019 18:50
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x.xx ml
Answers: 1

Chemistry, 21.06.2019 22:00
Afamily is one another name for a group on the table of elements.
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
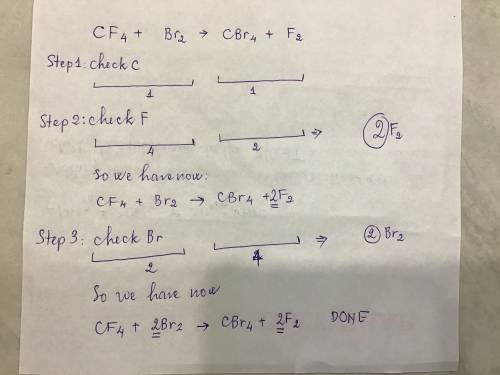
Balance this equation. _CF4+_Br2–_CBr4+_F2...
Questions

History, 23.11.2021 02:00

Computers and Technology, 23.11.2021 02:00

Mathematics, 23.11.2021 02:00


Social Studies, 23.11.2021 02:00


Mathematics, 23.11.2021 02:00

Chemistry, 23.11.2021 02:00



Engineering, 23.11.2021 02:00

Mathematics, 23.11.2021 02:00

English, 23.11.2021 02:00


Spanish, 23.11.2021 02:00


Mathematics, 23.11.2021 02:00

Mathematics, 23.11.2021 02:00

History, 23.11.2021 02:00

English, 23.11.2021 02:10