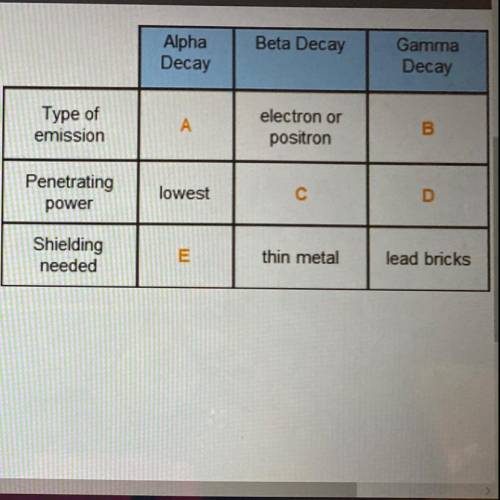
Fill in the blanks
Electron or positron
High-Energy photon
Particle with two prot...

Chemistry, 19.05.2020 03:10 erikloza12pdidtx
Fill in the blanks
Electron or positron
High-Energy photon
Particle with two protons and two neutrons

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 08:50
Why are enzymes important to cells? they bring about chemical reactions. they provide structural support. they form the two layers of membranes. they store large quantities of energy.
Answers: 2

Chemistry, 23.06.2019 12:30
The equilibrium constant kc for the reaction 2 nocl(g) → 2 no(g) + cl2(g) is 0.453 at a certain temperature. a mixture of nocl, no, and cl2 with concentrations 1.30, 1.20, and 0.600 m, respectively, was introduced into a container at this temperature. which of the following is true? 1. no apparent reaction takes place. 2. [cl2] = 0.30 m at equilibrium. 3. nocl(g) is produced until equilibrium is reached. 4. [nocl] = [no] = [cl2] at equilibrium. 5. cl2(g) is produced until equilibrium is
Answers: 3
You know the right answer?
Questions

Biology, 13.02.2020 01:09

Social Studies, 13.02.2020 01:12

English, 13.02.2020 01:12


Mathematics, 13.02.2020 01:12


Computers and Technology, 13.02.2020 01:13

Mathematics, 13.02.2020 01:13


Mathematics, 13.02.2020 01:13



Computers and Technology, 13.02.2020 01:14






Computers and Technology, 13.02.2020 01:14




