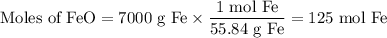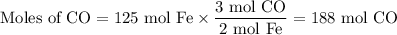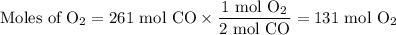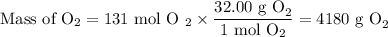Chemistry, 19.05.2020 02:17 mstrish71oteauw
Blast furnaces extra pure iron from the Iron(IIl)oxide in iron ore in a two step sequence. In the first step, carbon and oxygen react to form carbon monoxide:
2C(s)+O2(g) arrow 2CO(g)
In the second step, iron(lll) oxide and carbon monoxide react to form Iron and carbon dioxide:
Fe203(s) + 3CO(g) arrow 2Fe(s)+ 3CO2(g)
Suppose the yield of the first step is 71.% and the yield of the second step is 72.%. Calculate the mass of oxygen required to make 7.0 kg of iron.
Be sure your answer has a unit symbol, if needed, and is rounded to 2 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 12:00
Most materials are not magnetic because their magnetism has worn off. their magnetic domains are arranged randomly. they lack magnetic fields. earth’s heat has destroyed their magnetism.
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Blast furnaces extra pure iron from the Iron(IIl)oxide in iron ore in a two step sequence. In the fi...
Questions


Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01

History, 16.10.2020 08:01






Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01



History, 16.10.2020 08:01

History, 16.10.2020 08:01

History, 16.10.2020 08:01