
Urea (CH4N2O) is a common fertilizer that can be synthesized by the reaction of ammonia (NH3) with carbon dioxide as follows: 2NH3(aq)+CO2(aq)→CH4N2O(aq)+H2O(l) In an industrial synthesis of urea, a chemist combines 142.1 kg of ammonia with 211.4 kg of carbon dioxide and obtains 171.4 kg of urea. Part A Determine the limiting reactant. Express your answer as a chemical formula.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2


Chemistry, 23.06.2019 01:00
Reactions in cells take place at about a. 40°c b. 0° c. 100°c d. 60°c
Answers: 1

You know the right answer?
Urea (CH4N2O) is a common fertilizer that can be synthesized by the reaction of ammonia (NH3) with c...
Questions

Mathematics, 09.02.2021 09:10


Mathematics, 09.02.2021 09:10

Chemistry, 09.02.2021 09:10




Mathematics, 09.02.2021 09:10

History, 09.02.2021 09:10



Mathematics, 09.02.2021 09:10



History, 09.02.2021 09:10

History, 09.02.2021 09:10

Computers and Technology, 09.02.2021 09:10


English, 09.02.2021 09:10

Mathematics, 09.02.2021 09:10

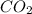
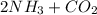 →
→ 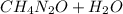
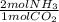 = 9,609 mol of NH3 present
= 9,609 mol of NH3 present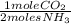 = 4,179.4 mol of CO2 present
= 4,179.4 mol of CO2 present

