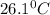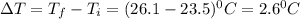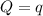Chemistry, 07.05.2020 05:16 rosepetals2938
The energy content of food is typically determined using a bomb calorimeter. Consider the combustion of a 0.22-g sample of butter in a bomb calorimeter having a heat capacity of 2.67 kJ/°C. If the temperature of the calorimeter increases from 23.5°C to 26.1°C, calculate the energy of combustion per gram of butter. Energy of combustion = kJ/g

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

You know the right answer?
The energy content of food is typically determined using a bomb calorimeter. Consider the combustion...
Questions


History, 02.10.2019 08:30




History, 02.10.2019 08:30

English, 02.10.2019 08:30

History, 02.10.2019 08:30

History, 02.10.2019 08:30

Chemistry, 02.10.2019 08:30

Mathematics, 02.10.2019 08:30



English, 02.10.2019 08:30

Biology, 02.10.2019 08:30


Social Studies, 02.10.2019 08:30



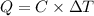
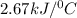
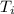 =
= 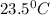
 =
=