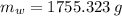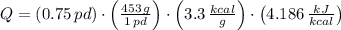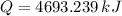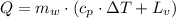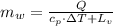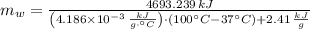Chemistry, 07.05.2020 05:12 marlesly87
The fuel value of hamburger is approximately 3.3 kcal/g. If a man eats 0.75 pounds of hamburger for lunch and none of the energy is stored in his body, estimate the amount of water that would have to be lost in perspiration to keep his body temperature constant. The heat of vaporization of water may be taken as 2.41 kJ/g.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 23.06.2019 05:00
110 g of water (specific heat = 4.184 j/g c) and 100 g of a metal sample (specific heat = 0.397 j/g c) are heated from 25 degrees c to 75 degrees c. which substance required more thermal energy?
Answers: 1
You know the right answer?
The fuel value of hamburger is approximately 3.3 kcal/g. If a man eats 0.75 pounds of hamburger for...
Questions

Health, 06.02.2021 22:50

Mathematics, 06.02.2021 22:50


Health, 06.02.2021 22:50

Mathematics, 06.02.2021 22:50



Chemistry, 06.02.2021 22:50



Mathematics, 06.02.2021 22:50

Mathematics, 06.02.2021 22:50


Mathematics, 06.02.2021 22:50



English, 06.02.2021 22:50



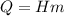
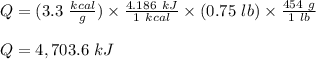
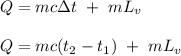
![Q = m[c(t_2 -t_1) \ + \ Lv]\\\\m = \frac{Q}{c(t_2 -t_1) \ + \ Lv} \\\\m = \frac{4,703.6 \times 10^3 }{4.186 (100-37) \ + \ 2.41 \times 10^3} \\\\m = 1,759.2 \ g\\\\m = 1.76 \ kg](/tpl/images/0651/6942/32919.png)