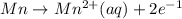A galvanic cell consists of one half-cell that contains Ag(s) and Ag+(aq), and one half-cell that contains Mn(s) and Mn2+(aq). What species are produced at the electrodes under standard conditions? Ag+(aq) + e- → Ag(s) E° = +0.80 V Mn2+(aq) + 2 e- → Mn(s) E° = -1.18 V A) Ag(aq) is formed at the cathode and, Mn(s) is formed at the anode. B) Ag(s) is formed at the cathode, and Mn2+(aq) is formed at the anode. C) Mn(s) is formed at the cathode, and Ag+(aq) is formed at the anode. D) Mn2+(aq) is formed at the cathode, and Ag(s) is formed at the anode.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Based on the equation and the information in the table, what is the enthalpy of the reaction?
Answers: 2

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 19:30
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1
You know the right answer?
A galvanic cell consists of one half-cell that contains Ag(s) and Ag+(aq), and one half-cell that co...
Questions



Computers and Technology, 01.06.2021 20:20



Mathematics, 01.06.2021 20:20

Chemistry, 01.06.2021 20:20


English, 01.06.2021 20:20

Biology, 01.06.2021 20:20


Social Studies, 01.06.2021 20:20

Mathematics, 01.06.2021 20:20

Biology, 01.06.2021 20:20

Mathematics, 01.06.2021 20:20

Mathematics, 01.06.2021 20:20


Social Studies, 01.06.2021 20:20


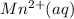 is formed at the anode.
is formed at the anode. 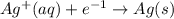 E=0.80 V
E=0.80 V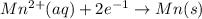 E=-1.18 V
E=-1.18 V