
Chemistry, 06.05.2020 02:37 DiamondW1258
If I add 375 of water to 125 mL of a 0.90 M NaOH solution, what will the molarity of the diluted solution be?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
If I add 375 of water to 125 mL of a 0.90 M NaOH solution, what will the molarity of the diluted sol...
Questions

English, 05.05.2020 04:35

Mathematics, 05.05.2020 04:35

English, 05.05.2020 04:35


Mathematics, 05.05.2020 04:36


Mathematics, 05.05.2020 04:36

Mathematics, 05.05.2020 04:36


Mathematics, 05.05.2020 04:36


Mathematics, 05.05.2020 04:36




Biology, 05.05.2020 04:36


Mathematics, 05.05.2020 04:36

Mathematics, 05.05.2020 04:36

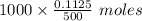 of NaOH or 0.225 moles of NaOH
of NaOH or 0.225 moles of NaOH

