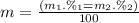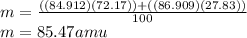Chemistry, 27.08.2019 07:00 Jerrikasmith28
Given that rubidium has two isotopes, 85rb and 87rb. calculate the average atomic mass of rubidium.
note that 85rb has an atomic mass of 84.912 amu and occurs at an abundance of 72.17% while 87rb has an atomic mass of 86.909 amu and occurs at an abundance of 27.83%.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:20
Both 1,2−dihydronaphthalene and 1,4−dihydronaphthalene may be selectively hydrogenated to 1,2,3,4−tetrahydronaphthalene. one of these isomers has a heat of hydrogenation of 101 kj/mol (24.1 kcal/mol), and the heat of hydrogenation of the other is 113 kj/mol (27.1 kcal/mol). match the heat of hydrogenation with the appropriate dihydronaphthalene.
Answers: 2

Chemistry, 21.06.2019 18:00
Which of the following is a compound? a.carbon b.oxygen c.hydrogen d.water
Answers: 2

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1
You know the right answer?
Given that rubidium has two isotopes, 85rb and 87rb. calculate the average atomic mass of rubidium.<...
Questions

Social Studies, 09.12.2021 02:30


Computers and Technology, 09.12.2021 02:30

SAT, 09.12.2021 02:30

Mathematics, 09.12.2021 02:30


Mathematics, 09.12.2021 02:30




Social Studies, 09.12.2021 02:30


Physics, 09.12.2021 02:30




Computers and Technology, 09.12.2021 02:30

SAT, 09.12.2021 02:30