Chemistry, 05.05.2020 00:44 Blakemiller2020
Using the following thermochemical equation, determine the amount of heat produced per kg of CO2 formed during the combustion of benzene (C6H6).
2 C6H6(l) + 15 O2(g) → 12 CO2(g) + 6 H2O(g) ΔH°rxn = -6278 kJ

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

Chemistry, 23.06.2019 01:30
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
You know the right answer?
Using the following thermochemical equation, determine the amount of heat produced per kg of CO2 for...
Questions


Biology, 20.12.2019 12:31

Chemistry, 20.12.2019 12:31


Business, 20.12.2019 12:31




Chemistry, 20.12.2019 12:31

Mathematics, 20.12.2019 12:31



Chemistry, 20.12.2019 12:31



Chemistry, 20.12.2019 12:31

History, 20.12.2019 12:31

Mathematics, 20.12.2019 12:31


Mathematics, 20.12.2019 12:31

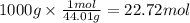
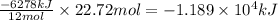
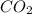 formed during the combustion of benzene
formed during the combustion of benzene 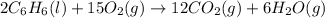
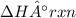 = -6278 kJ
= -6278 kJ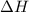 for the reaction comes out to be negative.
for the reaction comes out to be negative.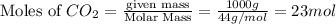 (1kg=1000g)
(1kg=1000g)