Chemistry, 03.05.2020 13:45 JamierW2005
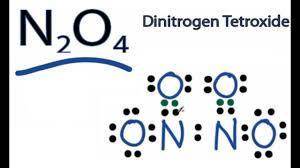
An oxide of nitrogen is 25.9% N by mass, has a molar mass of 108 g/mol, and contains no nitrogen-nitrogen or oxygen-oxygen bonds. Draw its Lewis structure (including all lone pair electrons) and name it. Include any nonzero formal charges in your structure.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2


You know the right answer?
An oxide of nitrogen is 25.9% N by mass, has a molar mass of 108 g/mol, and contains no nitrogen-nit...
Questions

Mathematics, 16.04.2020 23:31

Mathematics, 16.04.2020 23:31



Social Studies, 16.04.2020 23:31


English, 16.04.2020 23:31

Mathematics, 16.04.2020 23:31

Mathematics, 16.04.2020 23:31


History, 16.04.2020 23:31




Physics, 16.04.2020 23:32

Mathematics, 16.04.2020 23:32