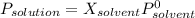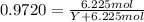Chemistry, 05.05.2020 04:23 NylaJohn29
The common laboratory solvent ethanol is often used to purify substances dissolved in it. The vapor pressure of ethanol , CH3CH2OH, is 54.68 mm Hg at 25 °C. In a laboratory experiment, students synthesized a new compound and found that when 32.83 grams of the compound were dissolved in 286.8 grams of ethanol, the vapor pressure of the solution was 53.15 mm Hg. The compound was also found to be nonvolatile and a non-electrolyte. What is the molecular weight of this compound? ethanol = CH3CH2OH = 46.07 g/mol.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:50
How do the energy differences between the higher energy levels of an atom compare with the energy difference between the lower energy level of the atom
Answers: 1

Chemistry, 21.06.2019 16:00
What is lincoln's purpose in writing this speech? question 1 options: to stress the difficulties of war to honor those who died in the war to call for an end to the war to call the country to join a new war
Answers: 1

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1
You know the right answer?
The common laboratory solvent ethanol is often used to purify substances dissolved in it. The vapor...
Questions

Physics, 16.01.2022 05:10


Mathematics, 16.01.2022 05:10


Biology, 16.01.2022 05:10

Mathematics, 16.01.2022 05:10


History, 16.01.2022 05:10

Mathematics, 16.01.2022 05:20

Geography, 16.01.2022 05:20

Mathematics, 16.01.2022 05:20

Biology, 16.01.2022 05:20

Mathematics, 16.01.2022 05:20

Social Studies, 16.01.2022 05:20

Mathematics, 16.01.2022 05:20

Mathematics, 16.01.2022 05:20



History, 16.01.2022 05:30

History, 16.01.2022 05:30