
Chemistry, 05.05.2020 06:28 ricksterv5000
Calcium carbide reacts with water to produce acetylene gas according to the following equation: CaC2(s) + 2H2O(l)C2H2(g) + Ca(OH)2(aq) The product gas, C2H2, is collected over water at a temperature of 25 °C and a pressure of 749 mm Hg. If the wet C2H2 gas formed occupies a volume of 5.87 L, the number of moles of CaC2 reacted was mol. The vapor pressure of water is 23.8 mm Hg at 25 °C. Submit Answer

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3


Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
Calcium carbide reacts with water to produce acetylene gas according to the following equation: CaC2...
Questions

English, 03.11.2020 01:00



Mathematics, 03.11.2020 01:00

English, 03.11.2020 01:00


English, 03.11.2020 01:00


History, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00

English, 03.11.2020 01:00

Social Studies, 03.11.2020 01:00





Mathematics, 03.11.2020 01:00

English, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00

History, 03.11.2020 01:00

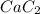 reacted was 0.229
reacted was 0.229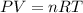
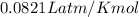
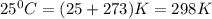
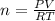
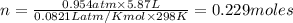
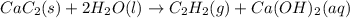
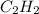 is produced by = 1 mole of
is produced by = 1 mole of 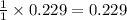 moles of
moles of 

