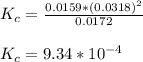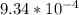Chemistry, 05.05.2020 07:20 stphdrn4347
Onsider the reversible dissolution of lead(II) chloride. P b C l 2 ( s ) − ⇀ ↽ − P b 2 + ( a q ) + 2 C l − ( a q ) PbClX2(s)↽−−⇀PbX2+(aq)+2ClX−(aq) Suppose you add 0.2393 g of P b C l 2 ( s ) PbClX2(s) to 50.0 mL of water. When the solution reaches equilibrium, you find that the concentration of P b 2 + ( a q ) PbX2+(aq) is 0.0159 M and the concentration of C l − ( a q ) ClX−(aq) is 0.0318 M. What is the value of the equilibrium constant, Kc, for the dissolution of P b C l 2 PbClX2?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2
You know the right answer?
Onsider the reversible dissolution of lead(II) chloride. P b C l 2 ( s ) − ⇀ ↽ − P b 2 + ( a q ) + 2...
Questions

Chemistry, 01.09.2019 11:50





Mathematics, 01.09.2019 11:50

History, 01.09.2019 11:50


Chemistry, 01.09.2019 11:50



Physics, 01.09.2019 11:50

Social Studies, 01.09.2019 11:50


Mathematics, 01.09.2019 11:50

Computers and Technology, 01.09.2019 11:50

Mathematics, 01.09.2019 11:50



Mathematics, 01.09.2019 11:50

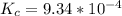
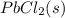 ⇌
⇌ 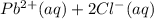
![[Pb^{2+}]](/tpl/images/0634/7278/0acfd.png) = 0.0159 M
= 0.0159 M
![[Cl^-]](/tpl/images/0634/7278/0726e.png) = 0.0318 M
= 0.0318 M
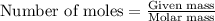
 Number of moles of PbCl₂
Number of moles of PbCl₂ 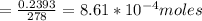
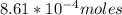
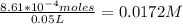
![K_c=\frac{[Pb^{2+}][Cl^-]}{[PbCl_2]} \\\\](/tpl/images/0634/7278/c4946.png)