Chemistry, 05.05.2020 07:10 ghanim1963
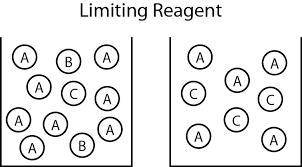
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution containing 5.76 g5.76 g of potassium chloride. Enter the balanced chemical equation for this reaction. Be sure to include all physical states. balanced chemical equation: Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s)+2KNO 3(aq)Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s) +2KNO3(aq) What is the limiting reactant? potassium chloride lead(II) nitrate The percent yield for the reaction is 87.5%87.5% . How many grams of the precipitate are formed? precipitate formed: gg How many grams of the excess reactant remain?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which one of the following gases is not an important component of soil?
Answers: 2

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1


You know the right answer?
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution cont...
Questions


Mathematics, 10.11.2020 22:50

Arts, 10.11.2020 22:50


Mathematics, 10.11.2020 22:50

Mathematics, 10.11.2020 22:50

Mathematics, 10.11.2020 22:50


Mathematics, 10.11.2020 22:50





Physics, 10.11.2020 22:50

Arts, 10.11.2020 22:50




Computers and Technology, 10.11.2020 22:50

Mathematics, 10.11.2020 22:50

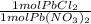 *
* 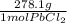 * 87.5/100 = 7.20 g PbCl₂
* 87.5/100 = 7.20 g PbCl₂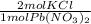 *
* 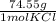 = 3.86 g KCl
= 3.86 g KCl