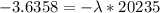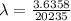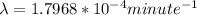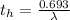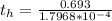Be sure to answer all parts. A freshly isolated sample of 90Y was found to have an activity of 2.2 × 105 disintegrations per minute at 1:00 p. m. on December 3, 2006. At 2:15 p. m. on December 17, 2006, its activity was measured again and found to be 5.8 × 103 disintegrations per minute. Calculate the half-life of 90Y. Enter your answer in scientific notation.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Be sure to answer all parts. A freshly isolated sample of 90Y was found to have an activity of 2.2 ×...
Questions




Mathematics, 07.07.2020 20:01


Computers and Technology, 07.07.2020 20:01









Mathematics, 07.07.2020 20:01

Mathematics, 07.07.2020 20:01





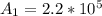 per minute
per minute 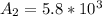 per minute
per minute 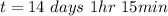
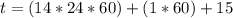
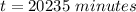
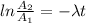
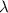 is the rate constant
is the rate constant ![ln [\frac{5.8 * 10^{3}}{2.2 *10^{5}} ] = - \lambda * 20235](/tpl/images/0634/6329/6ece6.png)