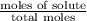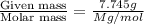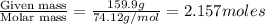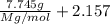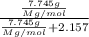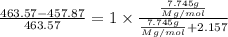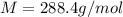The common laboratory solvent diethyl ether (ether) is often used to purify substances dissolved in it. The vapor pressure of diethyl ether , CH3CH2OCH2CH3, is 463.57 mm Hg at 25 °C. In a laboratory experiment, students synthesized a new compound and found that when 7.745 grams of the compound were dissolved in 159.9 grams of diethyl ether, the vapor pressure of the solution was 457.87 mm Hg. The compound was also found to be nonvolatile and a non-electrolyte. What is the molecular weight of this compound?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

You know the right answer?
The common laboratory solvent diethyl ether (ether) is often used to purify substances dissolved in...
Questions

Mathematics, 13.03.2021 01:00



History, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00

History, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00





Mathematics, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00



Mathematics, 13.03.2021 01:00



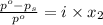
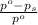 = relative lowering in vapor pressure
= relative lowering in vapor pressure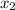 = mole fraction of solute =
= mole fraction of solute =