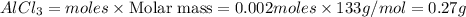Chemistry, 05.05.2020 17:25 morganhines181
Be sure to answer all parts. The balanced equation for the reaction of aluminum metal and chlorine gas is 2Al(s) + 3Cl2(g) → 2AlCl3(s) Assume that 0.80 g Al is mixed with 0.23 g Cl2. (a) What is the limiting reactant? Cl2 Al (b) What is the maximum amount of AlCl3, in grams, that can be produced? g AlCl3

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2

Chemistry, 22.06.2019 13:30
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1


Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2
You know the right answer?
Be sure to answer all parts. The balanced equation for the reaction of aluminum metal and chlorine g...
Questions



Biology, 27.01.2021 16:50

Advanced Placement (AP), 27.01.2021 16:50




Social Studies, 27.01.2021 16:50

Mathematics, 27.01.2021 16:50

Mathematics, 27.01.2021 16:50

Computers and Technology, 27.01.2021 16:50

English, 27.01.2021 16:50



Mathematics, 27.01.2021 16:50




Spanish, 27.01.2021 16:50

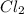 is the limiting reagent
is the limiting reagent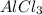 will be produced.
will be produced.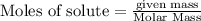
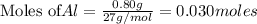
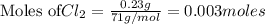
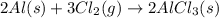
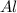
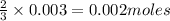 of
of