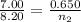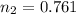Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 23:00
Which organism develops breathing organism develops breathing organs from pharyngeal arches? shark, spider, sea star, sea horse
Answers: 2

Chemistry, 23.06.2019 05:00
What is dhmo? hint: you find it everywhere something is wet..
Answers: 1

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1
You know the right answer?
3.00 L of Ch4 is known to contain 0.650 moles at a certain temperature and pressure if the volume of...
Questions

Biology, 01.06.2021 19:40

Arts, 01.06.2021 19:50

English, 01.06.2021 19:50


Mathematics, 01.06.2021 19:50


Social Studies, 01.06.2021 19:50

French, 01.06.2021 19:50




Mathematics, 01.06.2021 19:50

Mathematics, 01.06.2021 19:50


Mathematics, 01.06.2021 19:50

History, 01.06.2021 19:50

Mathematics, 01.06.2021 19:50

Mathematics, 01.06.2021 19:50

Geography, 01.06.2021 19:50

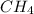 were added
were added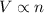
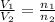
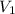 = initial volume of gas = 7.00 L
= initial volume of gas = 7.00 L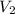 = final volume of gas = 8.20 L
= final volume of gas = 8.20 L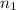 = initial moles of gas = 0.650 mole
= initial moles of gas = 0.650 mole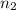 = final moles of gas = ?
= final moles of gas = ?