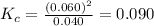Chemistry, 05.05.2020 18:04 AgentPangolin
At high temperatures, carbon reacts with O2 to produce CO as follows: C(s) O2(g) 2CO(g). When 0.350 mol of O2 and excess carbon were placed in a 5.00-L container and heated, the equilibrium concentration of CO was found to be 0.060 M. What is the equilibrium constant, Kc, for this reaction

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Estructura 7.2 completar repaso 1 - ¿lógico o ilógico? 1 - ¿lógico o ilógico? listen and indicate whether each question and response is lógico or ilógico.
Answers: 3

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 00:30
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 04:00
What three natural resources are found in the great lakes region
Answers: 2
You know the right answer?
At high temperatures, carbon reacts with O2 to produce CO as follows: C(s) O2(g) 2CO(g). When 0.350...
Questions



English, 26.10.2019 02:43


Mathematics, 26.10.2019 02:43



Mathematics, 26.10.2019 02:43




Chemistry, 26.10.2019 02:43

History, 26.10.2019 02:43

History, 26.10.2019 02:43

Computers and Technology, 26.10.2019 02:43

Computers and Technology, 26.10.2019 02:43




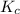 is 0.090.
is 0.090.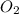 =
= 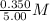 = 0.0700 M
= 0.0700 M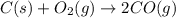
![K_{c}=\frac{[CO]^{2}}{[O_{2}]}](/tpl/images/0641/0273/4df57.png) , where [CO] and
, where [CO] and ![[O_{2}]](/tpl/images/0641/0273/9a638.png) represents equilibrium concentration of CO and
represents equilibrium concentration of CO and ![[CO]=2x=0.060](/tpl/images/0641/0273/522d6.png)