
Chemistry, 05.05.2020 17:57 mooneyhope24
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9.51 g N 2(g) and 1.96 g H2(g) react to produce NH
3(8)?
a.-29.8 kJ/mol-rxn
b.-43.7 kJ/mol-rxn
C.-10.3 kJ/mol-rxn
d.-20.7 kJ/mol-rxn
e. -65.6 kJ/mol-rxn

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3

Chemistry, 23.06.2019 07:00
In order for a high temperature boiler or steam engine to produce superheated water, or steam: the heat source must be greater than 100°c the water must be permitted to evaporate quickly the system must be sealed and become pressurized above atmospheric pressure the vapor pressure must be kept below 760 mm(hg)
Answers: 1
You know the right answer?
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9...
Questions





Social Studies, 11.03.2021 03:10

Mathematics, 11.03.2021 03:10

Mathematics, 11.03.2021 03:10

Chemistry, 11.03.2021 03:10



Chemistry, 11.03.2021 03:10


Mathematics, 11.03.2021 03:10

Mathematics, 11.03.2021 03:10


Mathematics, 11.03.2021 03:10

Mathematics, 11.03.2021 03:10



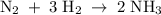
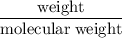
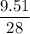
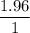
 2 moles of ammonia.
2 moles of ammonia.


